DESCRIPTION
Groups had to create board games that involved chemicals and physical changes. We needed to choose a theme for the game to revolve around. Using the knowledge about the chemical reactions we had learned in class, groups incorporated one of each kind of reaction into their board game. We were required to use at least 3 main reactions in each game, including a single replacement, double replacement, and a production of a gas, and also included the lighting of an LED, a chemical, and a physical/mechanical change of energy. We wrote detailed explanations of each reaction, blueprints, safety precautions, and the rules of the game.
My group's board game theme was battleship, and our game rules can be found below.
CONTENT
I've included some of the key concepts of the unit below.
Single replacement reaction- a type of chemical reaction where an element reacts with a compound and takes the place of another element in that compound
Double replacement reaction- a type of chemical reaction where two compounds react and the positive and negative ions of the reactants switch places
Cation- positive ion
Anion- negative ion
Compound- something composed of two more more elements
Aq- Aqueous, dissolves in water
S- Solid
L- Liquid
G- Gas
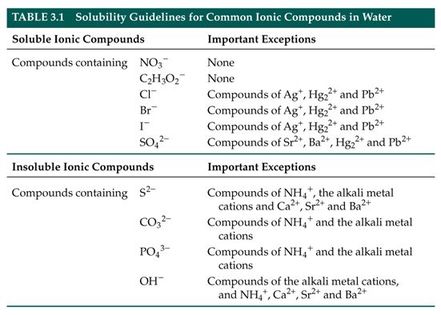
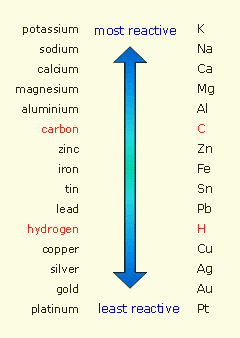
Below are images of the Solubility Guidelines and Reactivity Series.
The Solubility Guidelines will predict the state of the compounds in the reaction and will dictate what state the products of a chemical reaction will take. The Reactivity Series is used to determine the products of single displacement reactions, where metal A will replace another metal B in a solution if A is higher in the series.
I've included some of the key concepts of the unit below.
Single replacement reaction- a type of chemical reaction where an element reacts with a compound and takes the place of another element in that compound
Double replacement reaction- a type of chemical reaction where two compounds react and the positive and negative ions of the reactants switch places
Cation- positive ion
Anion- negative ion
Compound- something composed of two more more elements
Aq- Aqueous, dissolves in water
S- Solid
L- Liquid
G- Gas
Below are images of the Solubility Guidelines and Reactivity Series.
The Solubility Guidelines will predict the state of the compounds in the reaction and will dictate what state the products of a chemical reaction will take. The Reactivity Series is used to determine the products of single displacement reactions, where metal A will replace another metal B in a solution if A is higher in the series.
Below are the reactions we had used in our board game.
Single replacement reaction
Reaction in word form: Copper Chloride + Magnesium ------> Magnesium Chloride + Copper
Reaction chemical formula: CuCl2 + Mg —> MgCl2 + Cu
We had a breadboard with an LED set up, and used alligator clips to connect magnesium to the circuit. The player that won would pour copper chloride onto the magnesium and create a reaction that would light the LED. The Magnesium replaces Copper in the reaction because of the difference in the Reactivity series, the farther away the elements are from each other, the bigger the reaction will be.
Double replacement reaction
Reaction in word form: Potassium chloride + lead nitrate ---> lead chloride + potassium nitrate
Reaction chemical formula: 2KCl + Pb(NO3)2 ------> 2KNO3 + PbCl2
The player would put a few drops of both potassium chloride and lead nitrate onto a laminated sheet, and the solution would turn yellow. The combination formed a precipitate, following the solubility guidelines.
Production of a gas
Reaction in word form: sodium bicarbonate + acetic acid ---> carbon dioxide + water + sodium ion + acetate ion
Reaction chemical formula: NaHCO3 + CH3COOH → CO2 + H2O + Na + CH3COO
The player would put a few drops of acetic acid into a small container of sodium bicarbonate, which would create bubbles.
Our chemical change was the reaction between the magnesium and copper chloride, and having to pick up and move pieces around the board was our physical change of energy.
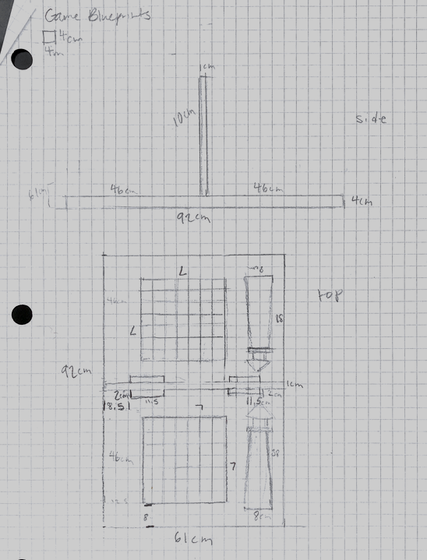
Images of our board game and blueprints can be found below.
Single replacement reaction
Reaction in word form: Copper Chloride + Magnesium ------> Magnesium Chloride + Copper
Reaction chemical formula: CuCl2 + Mg —> MgCl2 + Cu
We had a breadboard with an LED set up, and used alligator clips to connect magnesium to the circuit. The player that won would pour copper chloride onto the magnesium and create a reaction that would light the LED. The Magnesium replaces Copper in the reaction because of the difference in the Reactivity series, the farther away the elements are from each other, the bigger the reaction will be.
Double replacement reaction
Reaction in word form: Potassium chloride + lead nitrate ---> lead chloride + potassium nitrate
Reaction chemical formula: 2KCl + Pb(NO3)2 ------> 2KNO3 + PbCl2
The player would put a few drops of both potassium chloride and lead nitrate onto a laminated sheet, and the solution would turn yellow. The combination formed a precipitate, following the solubility guidelines.
Production of a gas
Reaction in word form: sodium bicarbonate + acetic acid ---> carbon dioxide + water + sodium ion + acetate ion
Reaction chemical formula: NaHCO3 + CH3COOH → CO2 + H2O + Na + CH3COO
The player would put a few drops of acetic acid into a small container of sodium bicarbonate, which would create bubbles.
Our chemical change was the reaction between the magnesium and copper chloride, and having to pick up and move pieces around the board was our physical change of energy.
Images of our board game and blueprints can be found below.
REFLECTION
Throughout this project, I had taken on a larger leadership role than I usually do. I usually work in groups where another member is quick to assume a leadership position, allowing me to involve myself in my own part instead of checking in with everyone. This time, there was nobody that wanted to take that role, so we tried to take turns sharing ideas and helping each other. My group had been easily distracted and it had been hard for us to get back on task and remain there for the rest of work time. I had easily gotten frustrated when things hadn't gone the way we'd planned. I'd been less empathetic in this group than I usually am, and could work to keep a more positive attitude. Communicating was difficult, and some of the group members had been absent during some parts of the building process, so we'd had to quickly update them the day they returned, but we were all still on different pages at times. We were good with time management, and were quick to decide on our ideas, although we could have spent more time planning ahead. When brainstorming ideas for the theme, we had decided that we wanted to do something original and had gone with the first idea we'd come up with. From there, we continued to act on the first ideas we'd come up with and altered them to better suit the game as time went on. We were happy with the end product, but could still spend more time thinking out the entire project next time.
I worked on this project with Henry Zhang, Tyler Marvier, and Trinity D'Aiello.
Throughout this project, I had taken on a larger leadership role than I usually do. I usually work in groups where another member is quick to assume a leadership position, allowing me to involve myself in my own part instead of checking in with everyone. This time, there was nobody that wanted to take that role, so we tried to take turns sharing ideas and helping each other. My group had been easily distracted and it had been hard for us to get back on task and remain there for the rest of work time. I had easily gotten frustrated when things hadn't gone the way we'd planned. I'd been less empathetic in this group than I usually am, and could work to keep a more positive attitude. Communicating was difficult, and some of the group members had been absent during some parts of the building process, so we'd had to quickly update them the day they returned, but we were all still on different pages at times. We were good with time management, and were quick to decide on our ideas, although we could have spent more time planning ahead. When brainstorming ideas for the theme, we had decided that we wanted to do something original and had gone with the first idea we'd come up with. From there, we continued to act on the first ideas we'd come up with and altered them to better suit the game as time went on. We were happy with the end product, but could still spend more time thinking out the entire project next time.
I worked on this project with Henry Zhang, Tyler Marvier, and Trinity D'Aiello.